NMPA published the twelve guidelines for in vitro diagnostic reagents related products on January 3, 2024. It comes as the File No.1 released by NMPA CMDE (Center for Medical Device Evaluation) in 2024.
Complete List
- Guideline on raw materials of in vitro diagnostic reagents
- Guideline on Brucella IgM/IgG Antibody Detection Reagents
- Guideline on Hepatitis C virus antibody detection reagent
- Guideline on Zika virus nucleic acid detection reagent
- Guideline on Aspergillus nucleic acid detection reagent
- Guideline on blood glucose monitoring system for self-testing
- Guideline on writing instructions for in vitro diagnostic reagents
- Guideline on tumor marker quantitative detection reagents
- Guideline on Influenza virus nucleic acid detection reagent
- Guideline on Influenza virus antigen detection reagent
- Guideline on pathogen-specific M-type immunoglobulin qualitative detection reagent
- Guideline on Hepatitis B virus DNA detection reagent
For our comprehensive review of “Guideline on raw materials of in vitro diagnostic reagents”, please visit HERE
For our recorded webinar of “Understanding IVD Reagents Registration Requirements: China & US Perspectives”, please visit HERE
For IVD classification draft catalog, click HERE
For the Guideline for IVD Overseas Clinical Data Acceptance, click HERE
- SEO Powered Content & PR Distribution. Get Amplified Today.
- PlatoData.Network Vertical Generative Ai. Empower Yourself. Access Here.
- PlatoAiStream. Web3 Intelligence. Knowledge Amplified. Access Here.
- PlatoESG. Carbon, CleanTech, Energy, Environment, Solar, Waste Management. Access Here.
- PlatoHealth. Biotech and Clinical Trials Intelligence. Access Here.
- Source: https://chinameddevice.com/ivd-guidelines/
- 1
- 2024
- acceptance
- antibody
- AS
- At
- b
- blood
- by
- catalog
- Center
- China
- classification
- click
- Clinical
- comes
- comprehensive
- data
- Detection
- device
- diagnostic
- dna
- draft
- Ether (ETH)
- evaluation
- File
- For
- guidelines
- HTTPS
- in
- Influenza
- instructions
- Issued
- IT
- January
- jpg
- marker
- materials
- medical
- medical device
- monitoring
- no
- of
- on
- once
- our
- overseas
- plato
- Plato Data Intelligence
- PlatoData
- please
- Products
- published
- qualitative
- quantitative
- Raw
- recorded
- Registration
- related
- released
- Requirements
- review
- system
- The
- tumor
- us
- virus
- Visit
- webinar
- writing
- zephyrnet
More from China Med Device
Two Imported Devices Gain Innovation Approval in December
Source Node: 2418669
Time Stamp: Dec 28, 2023
Ninety-Four Review Reports in 2022 Reveal NMPA’s Approval Process on Innovation & Priority Devices
Source Node: 1790781
Time Stamp: Dec 29, 2022
Three Imported Devices Found Defects in On-site Inspection
Source Node: 2415745
Time Stamp: Dec 19, 2023
Twenty-seven Devices Gain NMPA Innovation Status in Third Quarter 2023
Source Node: 2333997
Time Stamp: Oct 17, 2023
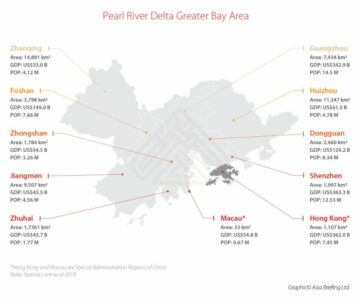
China Great Bay Area (GBA) Will be Impacted by New Hong Kong Medical Device Procurement & Market Access Acceleration
Source Node: 2205197
Time Stamp: Aug 9, 2023
More Class II & III Medical Devices Exempted from Clinical Trial
Source Node: 2190123
Time Stamp: Jul 27, 2023
Four Data Management Guidelines Issued for Devices and IVDs, Propelling China to be the Leader of Medical Information Technology
Source Node: 2271251
Time Stamp: Sep 14, 2023
15 Mandatory Standards to be Revised in 2023: Revisions Plan Asks Your Feedback
Source Node: 2093723
Time Stamp: May 12, 2023
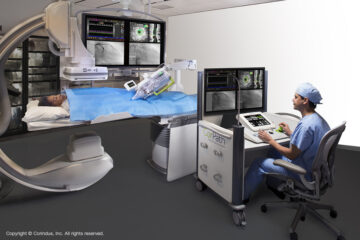
NMPA Review Report Released for Corindus’s Coronary Surgery Control Accessories
Source Node: 2134193
Time Stamp: Jun 14, 2023
2024 National Inspection Plan Released Today: Pay Attention to Your Device Standards
Source Node: 2521208
Time Stamp: Mar 20, 2024
Guideline on Human Factors Design Asks Second Round Feedback
Source Node: 2324793
Time Stamp: Oct 13, 2023