Medical Devices
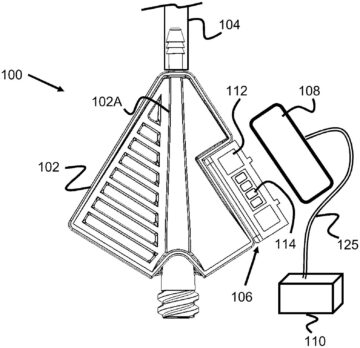
Enovis gets grant for real-time control of submerged arc welding power supply
Source Node: 2555376
Time Stamp: Apr 23, 2024
Revolutionising cardiac care: Caranx Medical makes history with robotic TAVI breakthrough
Source Node: 2554141
Time Stamp: Apr 22, 2024
Ujjain Medical Devices Park: Rs. 1,400 Crore Investment Proposal and 4,500 Job Opportunities! | Operon Strategist
Source Node: 2554139
Time Stamp: Apr 22, 2024
Exactech Strengthens Position as Soft Tissue Balancing Technology Leader with New Patents | BioSpace
Source Node: 2554135
Time Stamp: Apr 21, 2024
Hyalex Orthopaedics Reports Treatment of First Patients with Novel HYALEX® Knee Cartilage System | BioSpace
Source Node: 2554137
Time Stamp: Apr 21, 2024
Pantheon meets with FDA to discuss PMA submission for corneal implants
Source Node: 2551130
Time Stamp: Apr 19, 2024
Medtronic launches remote live streaming for surgical procedures
Source Node: 2551132
Time Stamp: Apr 19, 2024
Terumo gets grant for powered catheter system with magnetic attachment mechanism
Source Node: 2552415
Time Stamp: Apr 19, 2024
Fisher & Paykel Healthcare gets grant for patent granted for nasal seal for patient interface
Source Node: 2552417
Time Stamp: Apr 19, 2024
NMPA Review Report Released for Ivantis’ Hydrus Microstent
Source Node: 2551134
Time Stamp: Apr 19, 2024
CDSCO Test License for Medical Devices in India (Form MD 12, And MD 13) – Operon Strategist
Source Node: 2551128
Time Stamp: Apr 19, 2024
gaitQ and machineMD secure $1m to kickstart Parkinson’s movement study
Source Node: 2549917
Time Stamp: Apr 18, 2024
India Revolutionizes Drug & Medical Device Approval Process: Streamlined Path to Innovation | Operon Strategist
Source Node: 2549915
Time Stamp: Apr 18, 2024
Smith+Nephew announces new evidence supporting ALLEVYN LIFE Foam Dressing’s role in pressure injury prevention | BioSpace
Source Node: 2551106
Time Stamp: Apr 17, 2024