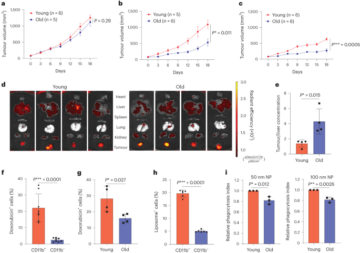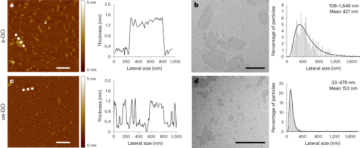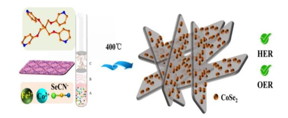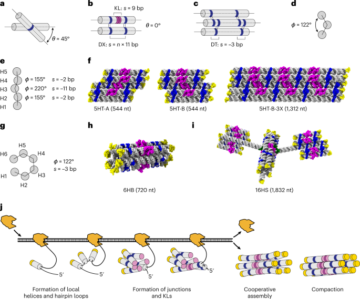
Greenough, L. et al. Adapting capillary gel electrophoresis as a sensitive, high-throughput method to accelerate characterization of nucleic acid metabolic enzymes. Nucleic Acids Res. 44, e15 (2016).
Farag, N. et al. Folding-upon-repair DNA nanoswitches for monitoring the activity of DNA repair enzymes. Angew. Chem. 133, 7359–7365 (2021).
Luo, X. & Hsing, I.-M. Immobilization-free electrochemical DNA polymerase assay. Electroanalysis 23, 923–926 (2011).
Boehr, D. D., Nussinov, R. & Wright, P. E. The role of dynamic conformational ensembles in biomolecular recognition. Nat. Chem. Biol. 5, 789–796 (2009).
Henzler-Wildman, K. & Kern, D. Dynamic personalities of proteins. Nature 450, 964–972 (2007).
Leveson-Gower, R. B., Mayer, C. & Roelfes, G. The importance of catalytic promiscuity for enzyme design and evolution. Nat. Rev. Chem. 3, 687–705 (2019).
Rotman, B. Measurement of activity of single molecules of β-d-galactosidase. Proc. Natl Acad. Sci. USA 47, 1981–1991 (1961).
Vogelstein, B. & Kinzler, K. W. Digital PCR. Proc. Natl Acad. Sci. USA 96, 9236–9241 (1999).
Rondelez, Y. et al. Microfabricated arrays of femtoliter chambers allow single molecule enzymology. Nat. Biotechnol. 23, 361–365 (2005).
Ono, T., Ichiki, T. & Noji, H. Digital enzyme assay using attoliter droplet array. Analyst 143, 4923–4929 (2018).
Guan, Z. et al. A highly parallel microfluidic droplet method enabling single-molecule counting for digital enzyme detection. Biomicrofluidics 8, 014110 (2014).
Rojek, M. J. & Walt, D. R. Observing single enzyme molecules interconvert between activity states upon heating. PLoS One 9, e86224 (2014).
Rissin, D. M. & Walt, D. R. Digital concentration readout of single enzyme molecules using femtoliter arrays and Poisson statistics. Nano Lett. 6, 520–523 (2006).
Liebherr, R. B. et al. Three-in-one enzyme assay based on single molecule detection in femtoliter arrays. Anal. Bioanal. Chem. 407, 7443–7452 (2015).
Obayashi, Y., Iino, R. & Noji, H. A single-molecule digital enzyme assay using alkaline phosphatase with a cumarin-based fluorogenic substrate. Analyst 140, 5065–5073 (2015).
Gorris, H. H., Rissin, D. M. & Walt, D. R. Stochastic inhibitor release and binding from single-enzyme molecules. Proc. Natl Acad. Sci. USA 104, 17680–17685 (2007).
English, B. P. et al. Ever-fluctuating single enzyme molecules: Michaelis–Menten equation revisited. Nat. Chem. Biol. 2, 87–94 (2006).
Hsin, T.-M. & Yeung, E. S. Single-molecule reactions in liposomes. Angew. Chem. Int. Ed. 46, 8032–8035 (2007).
Ueno, H., Kato, M., Minagawa, Y., Hirose, Y. & Noji, H. Elucidation and control of low and high active populations of alkaline phosphatase molecules for quantitative digital bioassay. Protein Sci. 30, 1628–1639 (2021).
Jiang, Y., Li, X. & Walt, D. R. Single-molecule analysis determines isozymes of human alkaline phosphatase in serum. Angew. Chem. Int. Ed. 59, 18010–18015 (2020).
Craig, D. B., Arriaga, E. A., Wong, J. C. Y., Lu, H. & Dovichi, N. J. Studies on Single alkaline phosphatase molecules: reaction rate and activation energy of a reaction catalyzed by a single molecule and the effect of thermal denaturation—the death of an enzyme. J. Am. Chem. Soc. 118, 5245–5253 (1996).
Sakuma, M. et al. Genetic perturbation alters functional substates in alkaline phosphatase. J. Am. Chem. Soc. 145, 2806–2814 (2023).
Gorris, H. H. & Walt, D. R. Mechanistic aspects of horseradish peroxidase elucidated through single-molecule studies. J. Am. Chem. Soc. 131, 6277–6282 (2009).
Ehrl, B. N., Liebherr, R. B. & Gorris, H. H. Single molecule kinetics of horseradish peroxidase exposed in large arrays of femtoliter-sized fused silica chambers. Analyst 138, 4260–4265 (2013).
Comellas-Aragonès, M. et al. A virus-based single-enzyme nanoreactor. Nat. Nanotechnol. 2, 635–639 (2007).
Liebherr, R. B., Renner, M. & Gorris, H. H. A single molecule perspective on the functional diversity of in vitro evolved β-glucuronidase. J. Am. Chem. Soc. 136, 5949–5955 (2014).
Jiang, Y. et al. Single-molecule mechanistic study of enzyme hysteresis. ACS Cent. Sci. 5, 1691–1698 (2019).
Watanabe, R., Sakuragi, T., Noji, H. & Nagata, S. Single-molecule analysis of phospholipid scrambling by TMEM16F. Biophys. J. 114, 558a (2018).
Tan, W. & Yeung, E. S. Monitoring the reactions of single enzyme molecules and single metal ions. Anal. Chem. 69, 4242–4248 (1997).
Sakakihara, S., Araki, S., Iino, R. & Noji, H. A single-molecule enzymatic assay in a directly accessible femtoliter droplet array. Lab Chip 10, 3355–3362 (2010).
Watanabe, R. et al. Arrayed lipid bilayer chambers allow single-molecule analysis of membrane transporter activity. Nat. Commun. 5, 4519 (2014).
Ueno, H., Sano, M., Hara, M. & Noji, H. Digital cascade assays for ADP- or ATP-producing enzymes using a femtoliter reactor array device. ACS Sens. 8, 3400–3407 (2023).
Noji, H., Minagawa, Y. & Ueno, H. Enzyme-based digital bioassay technology—key strategies and future perspectives. Lab Chip 22, 3092–3109 (2022).
Cox, K. J., Subramanian, H. K. K., Samaniego, C. C., Franco, E. & Choudhary, A. A universal method for sensitive and cell-free detection of CRISPR-associated nucleases. Chem. Sci. 10, 2653–2662 (2019).
Sternberg, S. H., Redding, S., Jinek, M., Greene, E. C. & Doudna, J. A. DNA interrogation by the CRISPR RNA-guided endonuclease Cas9. Nature 507, 62–67 (2014).
Montagne, K., Gines, G., Fujii, T. & Rondelez, Y. Boosting functionality of synthetic DNA circuits with tailored deactivation. Nat. Commun. 7, 13474 (2016).
Gines, G. et al. Isothermal digital detection of microRNA using background-free molecular circuit. Sci. Adv. 6, eaay5952 (2020).
Shenshin, V. A., Lescanne, C., Gines, G. & Rondelez, Y. A small-molecule chemical interface for molecular programs. Nucleic Acids Res. 49, 7765–7774 (2021).
Okumura, S. et al. Nonlinear decision-making with enzymatic neural networks. Nature 610, 496–501 (2022).
Li, Y. et al. Ultrasensitive isothermal detection of SARS-CoV-2 based on self-priming hairpin-utilized amplification of the G-rich sequence. Anal. Chem. 94, 17448–17455 (2022).
Richardson, C. D., Ray, G. J., DeWitt, M. A., Curie, G. L. & Corn, J. E. Enhancing homology-directed genome editing by catalytically active and inactive CRISPR-Cas9 using asymmetric donor DNA. Nat. Biotechnol. 34, 339–344 (2016).
Raper, A. T., Stephenson, A. A. & Suo, Z. Functional insights revealed by the kinetic mechanism of CRISPR/Cas9. J. Am. Chem. Soc. 140, 2971–2984 (2018).
Phaneuf, C. R. et al. Ultrasensitive multi-species detection of CRISPR-Cas9 by a portable centrifugal microfluidic platform. Anal. Methods 11, 559–565 (2019).
Zhang, X.-P. & Heyer, W.-D. in DNA Recombination: Methods and Protocols (ed. Tsubouchi, H.) 329–343 (Humana Press, 2011); https://doi.org/10.1007/978-1-61779-129-1_19
Tanford, C. in Advances in Protein Chemistry vol. 23 (eds. Anfinsen, C. B. et al.) 121–282 (Academic Press, 1968).
Berlett, B. S. & Stadtman, E. R. Protein oxidation in aging, disease, and oxidative stress. J. Biol. Chem. 272, 20313–20316 (1997).
Liu, G., Lin, Q., Jin, S. & Gao, C. The CRISPR-Cas toolbox and gene editing technologies. Mol. Cell 82, 333–347 (2022).
Phan, Q. A., Truong, L. B., Medina-Cruz, D., Dincer, C. & Mostafavi, E. CRISPR/Cas-powered nanobiosensors for diagnostics. Biosens. Bioelectron. 197, 113732 (2022).
Abate, A. R., Hung, T., Mary, P., Agresti, J. J. & Weitz, D. A. High-throughput injection with microfluidics using picoinjectors. Proc. Natl Acad. Sci. USA 107, 19163–19166 (2010).
Mazutis, L. & Griffiths, A. D. Selective droplet coalescence using microfluidic systems. Lab Chip 12, 1800–1806 (2012).
Mattox, A. K. et al. Applications of liquid biopsies for cancer. Sci. Transl. Med. 11, eaay1984 (2019).
Heitzer, E., Haque, I. S., Roberts, C. E. S. & Speicher, M. R. Current and future perspectives of liquid biopsies in genomics-driven oncology. Nat. Rev. Genet. 20, 71–88 (2019).
Abbotts, R. & Madhusudan, S. Human AP endonuclease 1 (APE1): from mechanistic insights to druggable target in cancer. Cancer Treat. Rev. 36, 425–435 (2010).
Collins, A. R. & Gaivão, I. DNA base excision repair as a biomarker in molecular epidemiology studies. Mol. Asp. Med. 28, 307–322 (2007).
Zaher, D. M. et al. Recent advances with alkaline phosphatase isoenzymes and their inhibitors. Arch. Pharm. 353, e2000011 (2020).
Sachsenhauser, V. & Bardwell, J. C. Directed evolution to improve protein folding in vivo. Curr. Opin. Struct. Biol. 48, 117–123 (2018).
Dramé-Maigné, A. et al. In vitro enzyme self-selection using molecular programs. ACS Synth. Biol. https://doi.org/10.1021/acssynbio.3c00385 (2024).
Xue, Q. & Yeung, E. S. Differences in the chemical reactivity of individual molecules of an enzyme. Nature 373, 681–683 (1995).
Craig, D. B. et al. Differences in the average single molecule activities of E. coli β-galactosidase: effect of source, enzyme molecule age and temperature of induction. J. Protein Chem. 22, 555–561 (2003).
Tawfik, D. S. Messy biology and the origins of evolutionary innovations. Nat. Chem. Biol. 6, 692–696 (2010).
Willensdorfer, M., Bürger, R. & Nowak, M. A. Phenotypic mutation rates and the abundance of abnormal proteins in yeast. PLoS Comput. Biol. 3, e203 (2007).
Yamagata, A., Masui, R., Kakuta, Y., Kuramitsu, S. & Fukuyama, K. Overexpression, purification and characterization of RecJ protein from Thermus thermophilus HB8 and its core domain. Nucleic Acids Res. 29, 4617–4624 (2001).
Menezes, R., Dramé-Maigné, A., Taly, V., Rondelez, Y. & Gines, G. Streamlined digital bioassays with a 3D printed sample changer. Analyst 145, 572–581 (2019).
Lobato-Dauzier, N. et al. Silicon chambers for enhanced incubation and imaging of microfluidic droplets. Lab Chip 23, 2854–2865 (2023).
Pekin, D. et al. Quantitative and sensitive detection of rare mutations using droplet-based microfluidics. Lab Chip 11, 2156–2166 (2011).
- SEO Powered Content & PR Distribution. Get Amplified Today.
- PlatoData.Network Vertical Generative Ai. Empower Yourself. Access Here.
- PlatoAiStream. Web3 Intelligence. Knowledge Amplified. Access Here.
- PlatoESG. Carbon, CleanTech, Energy, Environment, Solar, Waste Management. Access Here.
- PlatoHealth. Biotech and Clinical Trials Intelligence. Access Here.
- Source: https://www.nature.com/articles/s41565-024-01617-1
- ][p
- 003
- 05
- 1
- 10
- 11
- 114
- 12
- 13
- 14
- 15%
- 16
- 17
- 19
- 1995
- 1996
- 1999
- 2%
- 20
- 2001
- 2005
- 2006
- 2007
- 2009
- 2010
- 2011
- 2012
- 2013
- 2014
- 2015
- 2016
- 2017
- 2018
- 2019
- 2020
- 2021
- 2022
- 2023
- 2024
- 21
- 22
- 23
- 24
- 25
- 26
- 27
- 28
- 29
- 30
- 31
- 32
- 33
- 34
- 35%
- 36
- 375
- 39
- 3d
- 4
- 40
- 41
- 42
- 45
- 46
- 48
- 49
- 5
- 50
- 51
- 52
- 54
- 55
- 58
- 6
- 60
- 610
- 62
- 65
- 7
- 8
- 9
- a
- abnormal
- abundance
- academic
- accelerate
- accessible
- Activation
- active
- activities
- activity
- adapting
- Ads
- advances
- age
- Aging
- AL
- allow
- am
- Amplification
- an
- analysis
- and
- applications
- Array
- article
- AS
- aspects
- average
- b
- base
- based
- between
- binding
- biology
- biomarker
- boosting
- by
- Cancer
- cascade
- cent
- central
- Changer
- chemical
- circuit
- circuits
- click
- coalescence
- combining
- concentration
- control
- Core
- counting
- CRISPR
- Current
- Death
- Decision Making
- Design
- Detection
- determines
- device
- Diagnostics
- differences
- digital
- directed
- directly
- Disease
- Diversity
- dna
- domain
- dynamic
- e
- E&T
- ed
- effect
- enabling
- energy
- enhanced
- enhancing
- enzymatic
- equation
- Ether (ETH)
- evolution
- evolved
- exposed
- external
- For
- from
- functional
- functionality
- future
- GAO
- gene editing
- genetic
- genome
- harvard
- High
- highly
- http
- HTTPS
- human
- hung
- i
- Imaging
- importance
- improve
- in
- inactive
- INCUBATION
- individual
- induction
- innovations
- insights
- Interface
- ITS
- large
- li
- lin
- LINK
- Liquid
- Low
- mary
- measurement
- mechanism
- metabolic
- metal
- method
- methods
- molecular
- molecule
- monitoring
- Mutation
- nanotechnology
- Nature
- networks
- Neural
- neural networks
- NIH
- nonlinear
- observing
- of
- on
- oncology
- or
- origins
- oxidative
- Parallel
- PCR
- Personalities
- perspective
- perspectives
- platform
- plato
- Plato Data Intelligence
- PlatoData
- populations
- portable
- press
- programmable
- Programs
- Protein
- Proteins
- quantitative
- R
- RARE
- Rate
- Rates
- RAY
- reaction
- reactions
- reactor
- readout
- recent
- recognition
- reference
- release
- repair
- Revealed
- Role
- s
- sample
- SARS-CoV-2
- SBI
- Scholar
- SCI
- selective
- sensitive
- Sequence
- Serum
- Silicon
- single
- Source
- States
- statistics
- strategies
- streamlined
- stress
- studies
- Study
- substrate
- synthetic
- Systems
- T
- tailored
- Target
- Technologies
- The
- their
- thermal
- Through
- to
- Toolbox
- treat
- Universal
- upon
- using
- vivo
- W
- with
- wong
- Wright
- X
- zephyrnet