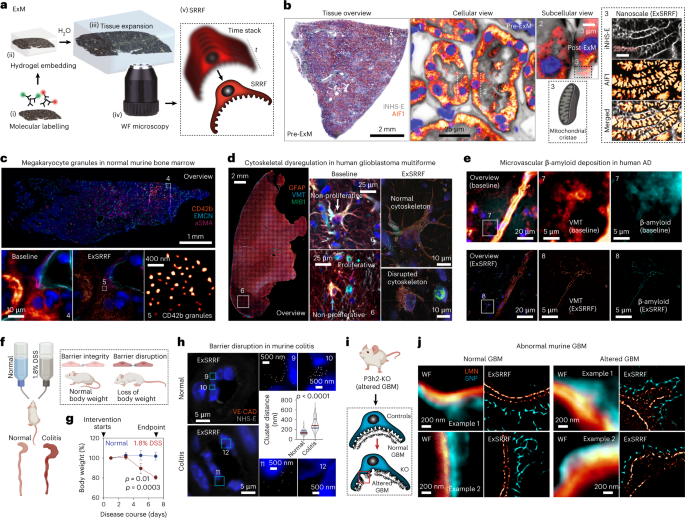
Human samples
FFPE tissues were collected according to institutional protocols. The control kidney specimens were collected in collaboration with the Division of Nephrology and Clinical Immunology, RWTH Aachen University. Kidney biopsies from patients with MCD and IgAN were obtained from the Hamburg Glomerulonephritis Registry (https://www.sfb1192.de/en/register). Ethical approvals were obtained from the Institutional Review Board of the RWTH Aachen University Medical Center (EK-016/17); the Ethik-Kommission der Ärztekammer Hamburg; and the local ethics committee of the Chamber of Physicians in Hamburg (PV4806). All of them are in accordance with the ethical principles stated by the Declaration of Helsinki.
In addition, specimens from patients with glioblastoma and AD were provided by the Institute of Neuropathology, University Medical Center Hamburg-Eppendorf. The study was reviewed by the ethics committee of the Hamburg Chamber of Physicians (WF72/17). A sample from human placenta was provided by the Division of Experimental Feto-Maternal Medicine, Department of Obstetrics and Fetal Medicine, University Medical Center Hamburg-Eppendorf, and approved by the Hamburg Chamber of Physicians (PV7312).
Murine samples
FFPE murine normal bone marrow was provided by the Department of Developmental Biology and Oncode Institute at Erasmus Medical Center Cancer Institute. Tissue collections were approved by the Animal Welfare/Ethics Committee of the EDC in accordance with legislation in the Netherlands (AVD1010020173387).
Control murine kidney tissue and experimental IRI tissue were collected in the Department of Medicine, University Medical Center Hamburg-Eppendorf. All the procedures were performed under the approval of the University Hospital Hamburg-Eppendorf and the Hamburg State Department for Animal Welfare under application no. N002/2020. Mouse anaesthesia: 30 min before surgery, 0.05 mg kg–1 buprenorphine was subcutaneously injected. At the time of surgery, narcosis was induced and maintained using isoflurane (5% isoflurane in pure O2 for induction, 2% isoflurane in O2 for maintenance). Induction of IRI: mice were anaesthetized and placed onto a heating pad (39 °C). The abdomen was opened via a median laparotomy and the renal artery of the right kidney was clamped for 30 min (micro-serrefine clamp, Fine Science Tools; 18055-03). The abdomen was filled with 0.9% NaCl and temporarily closed using Parafilm (Bemis). After this time, the clamp was removed, the abdomen was closed using a 3.0 suture and anaesthesia was stopped. Animals were placed back into their respective cages and had free access to soft chow and water, sweetened (eight drops of Natreen Classic) with added metamizole (1.3 mg ml–1, Ratiopharm). Endpoint: after 24 h, anaesthesia was induced again, and the abdomen was opened. Abdominal vessels were exposed, and the kidney perfusion was fixed via the abdominal aorta using 4% paraformaldehyde in 1× phosphate-buffered saline (PBS) at 200 mmHg for 5 min. The kidneys were harvested and stored in 4% PFA at 4 °C until further analysis.
Murine colon was obtained from controls and an experimental model of colitis. All the procedures were performed under the approval of the University Hospital Hamburg-Eppendorf and the Hamburg State Department for Animal Welfare under application no. TVA17/13. Mice were housed under specific pathogen-free conditions in individually ventilated cages with standard food and water ad libitum. Age- and sex-matched B6 wild-type mice between 8 and 12 weeks were used. During experiments, the mice were monitored daily. Intervention group mice were fed 1.8% DSS salt (MP Biomedicals; molecular weight, 36,000–50,000 Da) in drinking water for 7 days to induce colitis, whereas control mice were fed normal water41.
Archival tissues from an experimental model of disrupted GBM were obtained from a previous experiment. The generation of this mouse line was described in the original publication, together with ethical approval by Regierungspräsidium Freiburg (G16-122) and BGV Hansestadt Hamburg (Ü 004/2018).
De-waxing and antigen retrieval
Paraffin sections were cut at a thickness of 2–4 µm and mounted on Superfrost Plus slides. Then, all the samples were sequentially immersed in xylene 3× (10 min each) followed by an ethanol series (5 min each) of 3× 100%, 2× 70%, 1× 50% and finally 3× (5 min each) in double-deionized water. Antigen retrieval was performed with Agilent DAKO Target Retrieval Solution at pH 9 (catalogue no. S236884-2) in a Braun Multiquick FS20 steamer for 15 min, followed by cooling down to room temperature for 30 min. The sections were then incubated in Agilent wash buffer solution (catalogue no. K800721-2) for 15 min at room temperature.
Fluorescent immunolabelling
Samples were incubated with primary antibodies at concentrations according to vendor’s guidelines in Agilent antibody diluent solution (catalogue no. K800621-2) overnight at 4 °C, followed by three times washing for 5 min with Agilent wash buffer solution. Then, sections were incubated with appropriate secondary antibodies as well as directly conjugated primary antibodies at concentrations according to vendor’s guidelines with DAPI (Sigma-Aldrich D9542) at a final concentration of 1 μg ml–1 in Agilent antibody diluent solution for 1 h at room temperature and washed again three times for 5 min with Agilent wash buffer solution. Pan-protein staining was performed using NHS-ester (succinimidyl ester) conjugated with Alexa Fluor 555 (Invitrogen A20009) according to vendor’s guidelines. The following secondary antibodies were used: goat anti-guinea pig Alexa Fluor 488 (Invitrogen A11073), goat anti-guinea pig Alexa Fluor 555 (Invitrogen A21435), goat anti-mouse IgG1 Alexa Fluor 488 (Invitrogen A21121), goat anti-rabbit Atto 647N (Sigma-Aldrich MFCD06798562), goat anti-rat Alexa Fluor 555 (Invitrogen A21434) and streptavidin Alexa Fluor 555 conjugate (Invitrogen S21381). After immunostaining, the samples were mounted with ProLong Gold (Invitrogen P36930) for pre-expansion imaging.
Fluorescent in-situ hybridization
Fluorescent in-situ hybridization was performed in FFPE murine kidney samples using the RNAscope Multiplex Fluorescent Reagent Kit V2 Assay (catalogue no. 323100) according to the manufacturer’s guidelines and as previously described42. RNAscope probe NPHS-1 from Advanced Cell Diagnostics (catalogue no. 43357) was used to detect NPHS-1 mRNA and Opal 570 dye from Akoya Biosciences (catalogue no. OP-001003; dilution, 1:1,500) was applied for signal development.
Coverslip removal
Previously immunolabelled sections that were mounted and imaged were briefly immersed in xylene for 5 min at room temperature, after which the coverslip was carefully removed using a razor blade. The mounting media were washed off with PBS for 5 min at room temperature.
Primary antibodies
Primary antibodies and their respective species and dilutions used in this study are as follows: AIF1 (Cell Signaling Technology 5318; rabbit, 1:200), aSMA/FITC conjugate (Abcam F3777; mouse, 1:200), calreticulin (Abcam ab92516; rabbit, 1:200), CD42b (Abcam ab227669; rabbit, 1:200), collagen IV (Abcam ab6586; rabbit, 1:200), cytokeratin 8 (R&D Systems MAB3165; mouse IgG1, 1:200), endomucin (Santa Cruz Biotechnology sc-65495; rat IgG2a, 1:200), GFAP (Thermo Fisher Scientific 14-9892-82; mouse IgG1, 1:200), MIB1 (Abcam ab124929; rabbit, 1:200), nephrin (Progen GP-N2; guinea pig, 1:100), synaptopodin (Synaptic Systems 163 004; guinea pig, 1:100), β-amyloid (Thermo Fisher Scientific 715800; rabbit, 1:200), vimentin (Progen GP53; guinea pig, 1:200), vWF (Agilent A008229-2; rabbit, 1:200), laminin (Abcam ab11575; rabbit, 1:200) and CD144 VE-CAD (Thermo Fisher Scientific 14-1441-82; rat, 1:200).
Tissue expansion
Protocols for tissue expansion for enhanced optical resolution in thin sections have been previously described6. Briefly, immunolabelled and fluorescence in-situ hybridized sections underwent anchoring treatment with 0.1 mg ml–1 Acryloyl-X, 6-((acryloyl)amino)hexanoic acid, succinimidyl ester (Invitrogen A20770). On the other hand, no further anchoring treatment was required for fluorescence in-situ hybridized sections as the RNAscope Multiplex Fluorescent Reagent Kit V2 Assay utilizes Akoya Biosciences proprietary Tyramide signal amplification technology, resulting in the covalent deposition of OPAL dye molecules to nearby proteins. Acryloyl-X aliquots were dissolved in anhydrous DMSO at a concentration of 10.0 mg ml–1 for long-term storage at −20 °C and diluted in PBS to the final concentration of 0.1 mg ml–1 at room temperature for 12 h. Tissue sections were then embedded into a gelling solution consisting of 1× PBS, 2 M NaCl, 8.625% sodium acrylate (Sigma-Aldrich 408220), 2.500% acrylamide (Sigma-Aldrich A3553), 0.100% N–N′-methylenbis-(acrylamide) (Sigma-Aldrich 146072), 0.010% 4-hydroxy-2,2,6,6-tetramethyl-piperidin-1-oxyl (Sigma-Aldrich 176141), 0.200% N,N,N′,N′-tetramethylethylenediamine (Sigma-Aldrich T9281) and 0.200% ammonium persulfate (PanReac AppliChem, A1142). Embedded sections in the gelling solution were then incubated at 4 °C for 30 min to allow the penetration of gelling solution into the tissue. After that, gelling chambers, each consisting of two coverslips as spacers on either side of the tissue to prevent compression and a third coverslip on top of the tissue, were constructed around the tissue. In addition, sections were incubated in a humidified oven at 37 °C for 2 h to complete gelation. Next, the gelling chambers were removed, and specimens were incubated in 8 U ml–1 proteinase K (Sigma-Aldrich P2308) in a Tris/EDTA-based digestion buffer (50 mM Tris (pH 8), 25 mM EDTA, 0.5% Triton X-100 and 0.8 M NaCl) at 60 °C for 4 h. Following digestion, gel-embedded tissue sections were placed in double-deionized water at room temperature for 60 min to allow for isotropic expansion. After completing expansion, the tissues were removed from the double-deionized water and mounted in glass-bottom chamber slides (Ibidi µ-Slide two-well glass bottom; catalogue no. 80287) for subsequent imaging.
Nanoruler mounting and expansion
ExM-compatible nanorulers, equipped with acrydite groups to allow for covalent binding to the polymer matrix during gelation and biotin anchors to allow for immobilization on a bovine serum albumin (BSA)–biotin/NeutrAvidin surface, were custom ordered from GATTAquant DNA Nanotechnologies. First, to allow for the immobilization of nanorulers, glass slides were coated with BSA–biotin/NeutrAvidin. More precisely, the glass slides were first washed three times with 1,000 μl PBS, after which they were incubated with 200 μl Pierce BSA, biotinylated (Thermo Scientific 29130) at a concentration of 1 mg ml–1, dissolved in PBS, followed by washing another three times with 1,000 μl PBS. Tissues were then incubated with 200 μl NeutrAvidin solution (1 mg ml–1 NeutrAvidin Protein (Thermo Scientific 31000) dissolved in PBS) for 5 min. The NeutrAvidin solution was washed by applying three times 1,000 μl PBS, supplemented with 10 mM magnesium chloride. ExM-compatible nanoruler stock solutions were diluted in a ratio of 1:10 in PBS, supplemented with 10 mM magnesium chloride and then applied to BSA–biotin/NeutrAvidin-coated glass slides, incubated for 30 min at room temperature, followed by three washing steps with 1,000 μl PBS supplemented with 10 mM magnesium chloride. The slides were mounted in PBS supplemented with 10 mM magnesium chloride for pre-expansion imaging.
After pre-expansion image acquisition, coverslips were removed. In analogy to the tissue expansion protocol described above, the nanorulers were embedded in the ExM gelling solution and incubated at 4 °C for 30 min, followed by the construction of gelling chambers, after which the gelation step was performed by incubation in a humidified oven at 37 °C for 2 h. After completing gelation, gelling chambers were removed and as a digestion step, the nanorulers were denatured to allow for subsequent expansion by incubation in a 50% formamide (Sigma-Aldrich F9037) solution for 2 h at room temperature, followed by incubation at 4 °C for 12 h. The denatured nanorulers were then placed in double-deionized water at room temperature for 60 min to allow for isotropic expansion. After expansion, the samples were removed from the double-deionized water and mounted in glass-bottom chamber slides (Ibidi µ-Slide two-well glass bottom; catalogue no. 80287) for post-expansion imaging.
Imaging of nanorulers
Pre- and post-expansion LED-based WF imaging of nanorulers was performed using the THUNDER Imager 3D Live Cell and 3D Cell Culture (Leica Microsystems) fitted with a ×100 objective (numerical aperture (NA), 1.47). LED intensity and exposure times for each condition were systematically optimized. To allow for subsequent pre- and post-expansion SRRF processing, time-stacked images (with each time stack consisting of 50 images) were obtained. Pre- and post-expansion confocal imaging of the nanorulers was performed using the Zeiss LSM 800 confocal microscope with AiryScan using the optimized ×63 objective (NA, 1.4) at 12-fold digital zoom with subsequent AiryScan processing using the ZEN2.6 (blue edition) software.
Assessment of nanoruler PSF separation
To evaluate the separation of the nanoruler PSF after image acquisition, time-stacked imaging files from all the groups (120–25 nm, pre- and post-expansion) were first pseudonymized for further unbiased quantitative analysis by an independent observer without involvement in sample preparation and image acquisition. From these time stacks, 16–20 regions covering one nanoruler were randomly selected per group based on the raw imaging data, after which SRRF processing was performed. Both raw and SRRF-processed images then underwent supervised histogram adjustments to determine nanoruler PSF separation, which was defined as two fluorescent maxima that were clearly separated by at least one pixel without any signal.
Assessment of expansion factor and reporting
To evaluate the expansion factor, distances between the two fluorescent probes of the nanorulers were measured using the ImageJ2 Version 2.3.0/1.53q plot profile tool. A line positioned centrally through both fluorophores was drawn and the profile of pixel intensity along that line was generated. Thereafter, the distance between the two intensity maxima, depicting the fluorophores’ centres, was calculated. As in a previous report6 as well as a thorough validation of the expansion factor at the nanoscale (using nanorulers), for all the ExSRRF images, correlative pre- and post-expansion images of the region of interest (ROI) were obtained to reference post-expansion distances to pre-expansion biological distances, confirming the stability of the expansion factor at a micrometre scale.
Pre- and post-expansion imaging of tissues
Pre- and post-expansion LED-based WF imaging of tissues was performed using the THUNDER Imager 3D Live Cell and 3D Cell Culture (Leica Microsystems). Low-magnification pre- and post-expansion whole-tissue overviews were performed with a ×20 objective (NA, 0.40). High-magnification pre- and post-expansion images were obtained using multiple objectives, including a ×40 objective (NA, 1.10), ×63 objective (NA, 1.10) and ×100 objective (NA, 1.47) after optimizing the LED intensity and exposure times. To enable post-expansion SRRF processing, time stacks (each consisting between 20 and 200 images depending on the experimental requirements) were obtained for each ROI.
Time-stack image registration and movement correction
To correct potential movement during imaging, images within each time stack were first registered in Python 3 using scikit-image43 (https://scikit-image.org/) and image registration libraries (https://image-registration.readthedocs.io/en/latest/). Each image in the stack was separately aligned to the first image in the time stack using a reference channel containing our main structure of interest. In each image of the reference channel, the structures were first smoothed using a Gaussian filter with a standard deviation of 1. Then, the lower 90 percentile of the pixel values were set to 0, only keeping the upper 10 percentile of pixel values. Subsequently, the histogram of each image in the time stack was adjusted to match the reference histogram. Finally, the shift between the two images was obtained with the function chi2 shift from the image registration library, which uses the discrete Fourier transform upsampling method. This shift was finally removed from all the channels of the image, which were cropped and padded to retain the original size and were then reconstituted into an image stack.
SRRF and NanoJ-SQUIRREL
SRRF processing of raw data was performed using the Fiji imaging software Version 2.3.0/1.53q (Max Planck Institute of Molecular Cell Biology and Genetics) in combination with the NanoJ-SRRF plug-in.
For the pre- and post-expansion nanoruler SRRF processing, raw time-stacked files were processed using the following SRRF settings. Ring radius, 0.5; radiality magnification, 10; axes in ring; 8; temporal analysis; temporal radiality, average radiality; remove positivity constraint ‘disabled’; renormalize ‘disabled’; do gradient smoothing ‘disabled’; weighting, do intensity weighting ‘active’; do gradient weighting ‘disabled’; corrections, minimize SRRF patterning ‘active’; fast linearize SRRF ‘disabled’.
For all other tissue-based images, raw time-stacked files or histogram-adjusted files were processed using the following settings. Ring radius, 0.5–2.0; radiality magnification, 3–10 (depending on the desired magnification); axes in ring, 2–8 (depending on the desired magnification); temporal analysis; temporal radiality, average radiality; remove positivity constraint ‘disabled’; renormalize ‘disabled’; do gradient smoothing ‘active’; weighting, do intensity weighting ‘active’; do gradient weighting ‘disabled’; corrections, minimize SRRF patterning ‘active’; fast linearize SRRF ‘disabled’. For the processing of the inverted NHS-ester channel (Fig. 1b), the NHS-ester signal was first inverted using the ‘Invert’ function in Fiji, after which SRRF processing was performed (as described above). For the assessment of quality improvement by the image registration process, the super-resolution quantitative image rating and reporting of error locations (SQUIRREL) algorithm was used. Corresponding ROIs were selected in both raw and registered datasets. After being processed with the SRRF algorithm, the obtained super-resolution images were compared with the first image of each associated stack using the SQUIRREL algorithm. The generated error maps were used to visually highlight regions of high discrepancy, whereas resolution-scaled Pearson coefficients were utilized for the quantitative comparison between raw and registered datasets.
Adjusting image orientation for figure preparation
Pre-expansion and post-expansion images (raw and SRRF-processed images) were rotated using Fiji to obtain a similar orientation of the structures in the X and Y axes. This approach did not generate visual artefacts, although discrete rotation artefacts at the edges of the image in areas without the true signal cannot be excluded in all the cases.
Quantification of FP and SD width
To measure the SD width in human kidney biopsies stained with nephrin, the ExSRRF images first underwent automated thresholding using the ImageJ threshold tool in combination with the default algorithm and auto-adjusted. The SD width was then measured using the ImageJ plot profile tool. A line positioned at a 90° angle through the thresholded SD was drawn at multiple random positions along the SD followed by a plot profile analysis of the signal width. To measure the FP width in human kidney biopsies, ExSRRF images from nephrin-stained human kidney biopsy samples first underwent stepwise ImageJ-based processing for indirect morphological identification and segmentation of FPs. First, thresholding using the default algorithm in the auto-adjust mode was performed, followed by image inversion, and watershed segmentation, based on which further supervised segmentation was performed resulting in the identification of FPs separated by the SD. The FP width was then measured using the ImageJ plot profile tool by drawing a line at a 90° angle through the FP at multiple random positions followed by a plot profile analysis of the signal width.
Automated quantification of SD density and dilation
To quantify the SD density, a multistep process was developed in Python 3 using the scikit-image library43. First, the ROI was extracted, where the structures of interest are present. Then, the structures of interest were extracted using ridge detection and post-processing. Finally, the density of these structures was calculated within the extracted ROI. The steps and parameters in this process were optimized on two images from the IgAN samples and one image from the MCD samples and only then extended to all the other images. The results were first evaluated visually before checking the quantitative densities.
To extract the ROI, the image was first downsampled to 25% of its original size. The image was then thresholded to remove low-level noise and smoothed using a Gaussian filter with a standard deviation of 8, followed by Otsu thresholding. Mathematical morphology (binary filling of holes and binary closing) was applied next. Finally, all the connected areas were extracted and only those larger than a certain threshold (5,000 pixels) were retained. The resulting ROIs were upsampled again to the original image size.
SD segmentation was performed with the help of ridge detection using the Meijering ridge detector44. The resulting ridges were thresholded to only retain values larger than 0.2 and mathematical morphology (opening) was applied. SD densities were finally obtained as the ratio of pixels associated to the SD within the ROI. In addition, SD dilation was calculated in a similar fashion to trabecular thickness and spacing45, a commonly used plug-in for bone density analysis. Local spacing at a pixel of the image is the diameter of the greatest circle that fits within the space between ridges, and which is a part of the ROI and contains the point. The implemented ImageJ macro calculated the mean, median, standard deviation and area fraction of ridge spacing. For this study, we used the median per image.
Automated quantification of ER stress
To quantify the damage caused by IRI, a multistep process similar to the calculation of SD density and dilation was developed in Python 3 using the scikit-image library43. First, the ROI was extracted, where the structures of interest are present. Then, the structures of interest were extracted using ridge detection and post-processing. Finally, the density of these structures was calculated within the extracted ROI. The steps and parameters in this process were adapted from the calculation of SD density and dilation and optimized on one image from the control samples and one image from the IRI samples and only then extended to all the other images. The results were first evaluated visually before checking the quantitative densities.
To extract the ROI, the image was first downsampled to 25% of its original size. The image was smoothed using a Gaussian filter with a standard deviation of 8 and a Gaussian filter with a standard deviation of 10, followed by Otsu thresholding. Mathematical morphology (binary filling of holes) was applied next. Finally, all the connected areas were extracted and only those larger than a certain threshold (5,000 pixels) were retained. The resulting ROIs were upsampled again to the original image size.
Segmentation of calreticulin-based ER networks was performed with the help of ridge detection using the Meijering ridge detector44. The resulting ridges were thresholded to only retain values larger than 0.25; small objects with a size of less than 125 pixels were removed, and mathematical morphology (opening) was applied. The ER densities were finally obtained as the ratio of pixels associated to the ER within the ROI. In addition, ER dilation was calculated in a similar fashion to trabecular thickness and spacing45, a commonly used plug-in for bone density analysis. Local spacing at a pixel of the image is the diameter of the greatest circle that fits within the space between the ridges, and which is a part of the ROI and contains the point. The implemented ImageJ macro calculated the mean, median, standard deviation and area fraction of ridge spacing.
Evaluation of drift corrections
The registrations of time stacks were evaluated by taking into account the offset by which the frames had to be shifted during the registration process. For each frame in the time stack, the offset distance (2-norm) of the x and y offsets was calculated using the linear algebra function in the NumPy library (https://numpy.org/) using Python 3. These time-wise offsets were summarized in the mean offset and last offset, referring to the offset of the final frame, usually the largest offset in a time stack. For each time point, the mean structural similarity index measure (MSSIM) and mean squared error (MSE) were calculated using the scikit-image library43. These metrics were calculated for both original and registered frames with respect to the first frame (reference frame for the registrations), only taking into account the overlapping regions of the image to allow comparability. They were summarized in the mean and last MSSIMs, and the mean and last MSEs before and after registration for each time stack. The values after registration were subtracted from the ones before registration to measure the impact of registration. The MSSIM is larger for more similar structures, which means that a negative change indicates an improvement through registration, whereas the MSE is smaller for more similar images; thus, a positive change indicates improvement.
A standard deviation (s.d.) in the offset (based on 0.5 × s.d. from the mean) was used as the determining factor to split the time stacks into two groups: low drift (the vast majority) and the ones with more registration needed with offsets larger than 0.5 × s.d. were defined as high drift. The two groups were then compared in terms of the improvement achieved during registration. All the codes were implemented in Python 3.
Evaluation of resolution quantification for reliable segmentation
To evaluate the sharpness and suitability of the images to be used for further analysis of the structures, the raw ExM, ExSRRF and STED images were compared with the initial ridges detected on each image using the structural similarity index measure. A structural similarity index measure closer to 1 indicated better structural similarity between the images and ridges, thus indicating that there is better correspondence between the computed ridges and structures from the raw images.
Transmission electron microscopy
Murine kidney tissues were briefly fixed with 4.0% PFA and 2.5% glutaraldehyde in 0.1 M cacodylate buffer (pH 7.4) in situ at room temperature, and then dissected, removed and cut into pieces of 1 mm3 and fixed for 48 h in the same solution at 4 °C. The tissue blocks were contrasted using 1% OsO4 (Roth 7436.1) at room temperature for 1 h and 1% uranyl acetate (Polysciences 21447-25) in 70% ethanol at room temperature for 1 h. After dehydration, tissue blocks were embedded in epoxy resin (Durcopan ACM, Sigma-Aldrich 44611), and ultrathin sections of 50 nm thickness were cut using a Leica EM UC6 ultramicrotome (Leica Microsystems). The sections were imaged using a Zeiss 910 transmission electron microscope and analysed using ITEM software Version 5.2 (build 4768).
Human kidney biopsies were dissected according to standard operating procedures during diagnostic work up and were transferred from 4% formaldehyde into a cacodylate buffer together with sucrose for 10 min at 80 °C. Next, OsO4 was applied for 2 h, followed by washing in cacodylate buffer plus sucrose two times for 5 min. Subsequently, the sample was contrasted with uranyl acetate for 1 h. The specimen was then put into ethanol baths with rising ethanol concentrations for 5 min each, followed by methyl-tert-butylether twice for 5 min each, methyl-tert-butylether plus epoxide mixture (in 1:3 dilution). Afterwards, the specimens were embedded in an epoxide mixture at 60 °C for 48 h and then at 100 °C for 11.5 h. Semithin and ultrathin sections were cut on a Reichert Jung Ultracut E701704 microtome. Grids were purchased from Polyscience. The grids were then analysed using EM instruments (EM 109 and EM 902, Zeiss) equipped with digital EM cameras (Tröndle). One glomerulus from each case was analysed.
STED microscopy
Fluorescent labelling of the STED samples was performed as described above. The images were taken with an Abberior Instruments FACILITY LINE microscope equipped with an inverted IX83 microscope (Olympus), a ×60 oil objective (UPlanXApo ×60/1.42 oil, Olympus), using pulsed excitation lasers at 488, 561 and 640 nm and a pulsed STED laser operating at 775 nm, as well as an Abberior Expert Line four-channel easy3D STED instrument equipped with a 775 nm depletion beam (Abberior Instruments), a ×60 oil objective (Nikon ×60/1.4) and a QUAD beam scanner using a pulsed 640 nm diode beam, depleted with the 775 nm STED beam and detected with an avalanche photodiode with a front 685 ± 70 nm band-pass filter. All the acquisition operations were controlled by the Lightbox software Version 16.3.16118. Finally, the deconvolution (Richardson–Lucy) technique was used to improve the image quality.
3D-printed imaging chamber
NEXTERION coverslips (size, 110.000 × 74.000 ± 0.200 mm; thickness, 0.175 ± 0.020 mm; SCHOTT) were used for the mounting of expanded tissues that were too large to fit into the Ibidi µ-Slide two-well glass-bottom chambers. A custom-built mounting solution, including a frame, elastic cushion and lid for the frame were printed using a 3D printer to carry the NEXTERION coverslips to allow for mounting in the THUNDER Imager 3D Live Cell and 3D Cell Culture (Leica Microsystems).
The frame carries the NEXTERION coverslip allowing for insertion in the THUNDER Imager 3D Live Cell and 3D Cell Culture (Leica Microsystems). The elastic cushion is placed between the NEXTERION coverslip and lid, preventing the cover glass from breaking when it is locked in place by the lid. The lid for the frame stabilizes the NEXTERION coverslip to the frame. The NEXTERION coverslip and cushion are fully locked to the frame by setting those wedges into notches in the frame. Tinkercad (Autodesk; https://www.tinkercad.com) was utilized to create designs for all the 3D-printed parts.
All 3D-printed parts and design features are shown in Extended Data Figs. 2 and 3. The frame and lid were printed using PolyLite PLA filament 1.75 mm (Polymaker). The cushion was printed using NinjaFlex TPU filament 1.75 mm (NinjaTek). Ender-5 Plus (Creality) was utilized for 3D printing. The settings used in Ultimaker Cura (v. 4.13.1; Ultimaker) are as follows. Nozzle size, 0.40 mm; layer height, 0.16 mm (PLA) and 0.20 mm (TPU); wall thickness, 1.20 mm (PLA) and 0.80 mm (TPU); top/bottom thickness, 1.20 mm (PLA) and 0.80 mm (TPU); nozzle temperature, 220 °C (PLA) and 235 °C (TPU); bed temperature, 50 °C; fan speed, 100%; print speed, 50 mm s–1 (PLA) and 20 mm s–1 (TPU); first-layer print speed, 25 mm s–1 (PLA) and 10 mm s–1 (TPU); infil, 10% and zigzag; build-plate adhesion, none. Masking tapes were used to create an adhesive surface on the bed.
Statistical analyses
All the statistical analyses were performed using GraphPad Prism (v. 9.3.1). Violin plots report the median and interquartile range. Significance was evaluated using the unpaired t-tests with Welch’s correction comparing two continuous variables; a paired t-test for before/after settings; and the Brown–Forsythe, Welch ANOVA and Dunnett’s tests when comparing three continuous variables. Receiver operating characteristic curves were generated using the Wilson/Brown method (95% confidence interval; results expressed as percentages). A p value below 0.05 is considered to be statistically significant.
- SEO Powered Content & PR Distribution. Get Amplified Today.
- Platoblockchain. Web3 Metaverse Intelligence. Knowledge Amplified. Access Here.
- Source: https://www.nature.com/articles/s41565-023-01328-z
- :is
- ][p
- $UP
- 000
- 1
- 1.3
- 10
- 100
- 11
- 110
- 2%
- 2012
- 2014
- 2017
- 39
- 3d
- 3D Printing
- 7
- 70
- 8
- 9
- 95%
- a
- above
- access
- According
- Account
- achieved
- ACM
- acquisition
- Ad
- adapted
- added
- addition
- Adjusted
- adjustments
- advanced
- After
- AL
- Alexa
- algorithm
- aligned
- All
- Allowing
- Although
- Amplification
- analysis
- Anchor
- and
- animal
- animals
- Another
- Antibodies
- Application
- applied
- Applying
- approach
- appropriate
- approval
- approved
- ARE
- AREA
- areas
- around
- AS
- assessment
- associated
- At
- autodesk
- Automated
- Avalanche
- average
- AXES
- back
- based
- BE
- Beam
- before
- being
- below
- Better
- between
- binding
- biology
- biotechnology
- BLADE
- Blocks
- Blue
- board
- BONE
- Bottom
- Breaking
- briefly
- BSA
- buffer
- build
- by
- calculated
- cameras
- Cancer
- cannot
- carefully
- carry
- case
- cases
- caused
- Center
- certain
- Chamber
- change
- Channel
- channels
- characteristic
- checking
- Circle
- clamp
- classic
- clearly
- click
- Clinical
- closed
- closer
- closing
- Codes
- collaboration
- collections
- COM
- combination
- committee
- commonly
- COMP
- compared
- comparing
- comparison
- complete
- completing
- concentration
- condition
- conditions
- confidence
- connected
- considered
- Consisting
- construction
- contains
- continuous
- control
- controlled
- controls
- Corrections
- Corresponding
- COVALENT
- cover
- covering
- create
- Culture
- custom
- Custom-built
- Cut
- DA
- daily
- data
- datasets
- Days
- Default
- defined
- density
- Department
- Depending
- depicting
- described
- Design
- designs
- detected
- Detection
- Determine
- determining
- developed
- Development
- developmental
- deviation
- DID
- digital
- dilution
- directly
- discrepancy
- distance
- Division
- dna
- down
- drawing
- drawn
- Drops
- during
- e
- each
- edition
- either
- embedded
- enable
- Endpoint
- enhanced
- equipped
- error
- Ether (ETH)
- ethical
- ethics
- evaluate
- evaluated
- excluded
- expanded
- expansion
- experiment
- expert
- exposed
- Exposure
- expressed
- extract
- Facility
- fan
- Fashion
- FAST
- Features
- Fed
- Fig
- Figure
- Files
- filled
- filter
- final
- Finally
- fine
- First
- fit
- fixed
- fluctuations
- followed
- following
- follows
- food
- For
- formaldehyde
- fps
- fraction
- FRAME
- Free
- from
- front
- fully
- function
- further
- generate
- generated
- generation
- Genetics
- glass
- Gold
- greatest
- Group
- Group’s
- guidelines
- hand
- Have
- height
- help
- High
- Highlight
- Holes
- Hospital
- HTTPS
- human
- Identification
- image
- images
- Imaging
- immersed
- Impact
- implemented
- improve
- improvement
- in
- Including
- incubated
- INCUBATION
- independent
- index
- indicated
- indicates
- Individually
- initial
- Institute
- Institutional
- instrument
- instruments
- interest
- intervention
- inversion
- involvement
- IT
- ITS
- keeping
- kidney
- Kidneys
- kit
- labelling
- large
- larger
- largest
- laser
- lasers
- Last
- layer
- Led
- Legislation
- libraries
- Library
- Line
- LINK
- live
- local
- locations
- locked
- long-term
- Low
- Macro
- Main
- maintenance
- Majority
- Maps
- Match
- mathematical
- Matrix
- max
- means
- measure
- Media
- medical
- medicine
- method
- Metrics
- mice
- Microscope
- Microscopy
- minimize
- mixture
- Mode
- model
- MOL
- molecular
- monitored
- more
- movement
- mRNA
- multiple
- Nature
- needed
- negative
- Netherlands
- networks
- New
- next
- NIH
- Noise
- normal
- novel
- numpy
- objective
- objectives
- objects
- obtain
- obtained
- of
- offset
- Oil
- olympus
- on
- ONE
- opened
- opening
- operating
- Operations
- optical
- optimized
- optimizing
- original
- Other
- overnight
- overview
- pad
- paired
- parameters
- part
- parts
- pathology
- patients
- PBS
- pearson
- percentages
- pieces
- Pixel
- Place
- platform
- plato
- Plato Data Intelligence
- PlatoData
- plus
- Point
- polymer
- positioned
- positions
- positive
- Positivity
- potential
- precisely
- present
- prevent
- preventing
- previous
- previously
- primary
- principles
- probe
- procedures
- process
- processing
- Profile
- profiling
- ProGen
- proprietary
- Protein
- Proteins
- protocol
- protocols
- provided
- Publication
- purchased
- put
- Python
- quality
- quantitative
- R&D
- Rabbit
- random
- range
- RAT
- rating
- ratio
- Raw
- raw data
- region
- regions
- registered
- Registration
- registry
- reliable
- remove
- Removed
- report
- Reporting
- required
- Requirements
- Resin
- Resolution
- respect
- respective
- resulting
- Results
- retain
- review
- reviewed
- Ring
- rising
- RNA
- ROI
- Room
- s
- salt
- same
- Santa
- Scale
- Science
- SD
- secondary
- sections
- segmentation
- selected
- separately
- Series
- Serum
- set
- setting
- settings
- shift
- shown
- Signal
- significance
- significant
- similar
- Size
- Slides
- small
- smaller
- Soft
- Software
- solution
- Solutions
- Space
- specific
- speed
- split
- Squared
- Stability
- stack
- Stacks
- standard
- State
- State Department
- stated
- statistical
- Step
- Steps
- stock
- stopped
- storage
- stored
- structural
- structure
- Study
- subsequent
- Subsequently
- suitability
- Surface
- Surgery
- Systems
- taking
- Target
- Technology
- terms
- tests
- that
- The
- the Netherlands
- their
- Them
- These
- Third
- three
- three-dimensional
- threshold
- Through
- time
- times
- tissues
- to
- together
- too
- tool
- tools
- top
- Tracing
- transferred
- Transform
- treatment
- Triton
- true
- Twice
- under
- university
- usually
- utilized
- utilizes
- validation
- value
- Values
- variables
- Vast
- version
- via
- Wall
- Water
- Weeks
- weight
- Welfare
- WELL
- which
- with
- within
- without
- Work
- zephyrnet
- Zhao
- zoom